Vein Enation-Woody Gall
HISTORY, DISTRIBUTION AND IMPORTANCE
Citrus vein enation disease was first described in California on rough lemon and Mexican lime and shown to be caused by an aphid-transmitted pathogen (Wallace & Drake, 1953; 1959). Woody gall was first described on rough lemon in Australia (Fraser, 1958; 1959), and it was soon demonstrated that these two symptoms were caused by the same agent (Wallace & Drake, 1962). Because the vein enation symptoms were the first ones described, this name is applied to the causal agent, but the major field symptoms are the galls on tree trunks and branches, and in these cases, woody gall disease is appropriate.
Vein enation/woody gall has been reported from Australia, Argentina, Brazil, China, Fiji, India, Indonesia, Iran, Japan, Kenya, Kiribati, Libya, Mauritius, Nepal, New Zealand, Papua New Guinea, Peru, Philippines, Reunion, Spain, South Africa, Tanzania, Turkey and USA (California). It probably occurs elsewhere, but field symptoms may be absent due to non-use of gall forming varieties and/or climatic conditions. Studies in California has shown that cooler coastal regions are more favorable to gall formation, and galled plants raised in a cool greenhouse and transplanted to the field develop few new galls and some old galls became smaller.
It is a non-destructive disease, and the only report of reduced growth is in young trees with extensive woody gall symptoms in Peru (Bazan de Segura & Ferrand, 1969).
NAME OF DISEASE AND SYNONYMS
Vein enation refers to the small outgrowths on the veins on the undersurface of the leaves of some citrus species. Woody gall describes the galls that form on the trunks and branches of some infected species.
Abbreviation: VE-WG
SYMPTOMOLOGY
General aspect of affected field tree
Normal.
Symptoms on trunk (on rootstock and/or scion), limbs and shoots
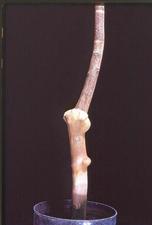
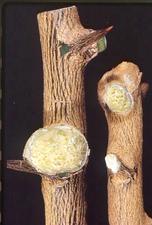
The first sign on susceptible citrus is usually a slight swelling of a small area of bark, often, but not always, near thorns or at the base of small twigs. The swellings develop a slight roughening and a light grey color. They enlarge slowly into irregularly-shaped galls (VE-WG. 01). Individual galls can grow into each other forming large masses of tumor-like tissue consisting of somewhat disorganized xylem tissue covered by fairly normal bark (VE-WG. 02). Frequently many sprouts develop on the galls (VE-WG. 03). Wounding on rough lemon and lime, such as from needle punctures, can initiate gall development (VE-WG. 04) (Wallace & Drake, 1961). Galls will frequently develop where there is no apparent wounding, but the regular appearance of galls adjacent to thorns and twigs appears to be associated with undeveloped thorn-like structures beneath the bark (VE-WG. 05), which resemble structures in non-infected plants (VE-WG. 06). Enations can form on thorns. On rough lemon rootstocks, woody galls appear on the rootstock (VE-WG. 07), and can encircle the budunion (VE-WG. 08). The cambial surface of gall tissue has a pitted appearance with numerous small holes (VE-WG. 09).
Symptoms on leaves
Projections or papillae on lateral veins and veinlets on the underside of leaves (VE-WG. 10) with corresponding depressions on the upper surface (VE-WG. 11). The enations range from slight swellings to 1-mm projections. The number per leaf varies from a few to many, and not all leaves on a plant will develop them.
Symptoms on fruit
None.
Histological and cytological symptoms
Enations are initiated as cytological abnormalities of phloem fiber primordial cells adjacent to protophloem sieve tubes (Hooper & Schneider, 1969). They enlarge by hyperplasia of the affected fiber primordials. The mesophyll and epidermal tissue on the abaxial side of leaf veins divide less prolifically and their growth ceases as leaves mature. Woody galls develop from affected cells of procambial tissue between metaxylem and metaphloem of vascular bundles, and contain large amounts of abnormal xylem tissue. Gall growth is indeterminate.
Interactions with other pathogens
There is a reported synergistic effect between CVEV and the yellow vein pathogen resulting in a marked enhancement of the yellow vein symptom in Etrog citron, rough lemon and Mexican lime (Weathers, 1960) (VE-WG. 12). In Japan, CVEV was shown to cross-protect against Citrus tristeza virus in some hosts (Koizumi & Sasaki, 1980). Vein enations are suppressed in Mexican lime and Pineapple sweet orange by CTV T30 isolates, but not by other isolates, and are not suppressed in sweet orange by viroids (Vidalakis et al., 2004).
CAUSAL AGENT: DESCRIPTION AND PROPERTIES
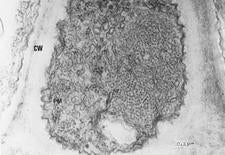
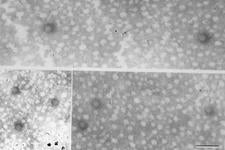
The graft and aphid-transmissibility of the agent strongly suggested that it is a virus, and the abbreviation CVEV has been adopted. Virus-like particles have been observed by electron microscopy of enations from rough lemon leaves (VE-WG. 13) and in the salivary glands of infectious aphids (Maharaj & da Graca, 1988). Isometric particles have been purified (VE-WG. 14), and based on morphology and disease characteristics, it was suggested that CVEV might be a luteovirus (da Graça & Maharaj, 1991). It was then demonstrated that positive ELISA results could be obtained for CVEV using some commercial Barley yellow dwarf virus kits (Clark & da Graça, 2000), and that isometric particles could be trapped by the antiserum for electron microscopy. Attempts to amplify cDNA using luteovirus primers have not been successful, thus the luteovirus proposal remains unconfirmed.
Strains – none described
HOST RANGE
Restricted to Citrus spp. No non-rutaceous hosts known.
Sensitive species, varieties or combinations
Rough lemon, Volkamer lemon, Rangpur lime and Mexican lime develop galls. Lime and sour orange develop conspicuous vein enations in the field, while sweet orange, lemon, mandarin and rough lemon develop enations under cool greenhouse conditions. Enations are sometimes present in infected sweet orange, lemon, mandarin, Palestine sweet lime, and kumquat. In India, enations have been observed on grapefruit, C. amblycarpa, C. macroptera, C. latipes and C. pennivesculata (Mali et al., 1976).
Symptomless species, varieties or combinations
- Immune – none described
- Tolerant (symptomless carrier) – apart from India (see above), grapefruit appears to be a symptomless carrier elsewhere.
- No specific reports on other Citrus spp. or relatives.
TRANSMISSION
Natural
CVEV is transmitted in a persistent manner by several species of aphids, namely Myzus persicae, Aphis gossypii, Toxoptera aurantii (Hermoso de Mendoza, 1993) and T. citricida (Maharaj & da Graca, 1989), which are also semi-persistent vectors of CTV. There is a latency period of 2-3 days. There is no evidence for seed transmission.
Experimental
Graft inoculation with buds and dodder transmission.
EPIDEMIOLOGY
Not studied
DIAGNOSIS
Diagnostic field symptoms
Woody galls on stems and trunks, and vein enations on leaves of citrus species listed under Host Range.
Comparison with other diseases
Woody galls of unknown etiology have been reported on sweet orange (VE-WG. 15), and insect damage appears to cause outgrowths on uninfected leaves (VE-WG. 16).
Biological indexing
- Indicator plants and inoculum: Mexican lime and sour orange seedlings. Inoculate with two buds. Alternative indicator: rough lemon, but enations can be smaller. Greenhouse temperatures should be 24-27C max. day/18-21C min. night.
- Observed symptoms: vein enations appear on new leaves. Initially only some leaves develop one or several enations, but later they increase in number. Woody galls develop on stems, and they show up on Mexican lime several weeks after the enations appear. Gall development on rough lemon and Volkamer lemon is more extensive.
- Incubation and development of symptoms: enations usually appear after 5-8 weeks. Galls appear several weeks after enations; more extensive on rough and Volkamer lemons than on Mexican lime.
- A shock reaction in Mexican lime consisting of leaf droop is used in Spain as a diagnostic symptom (VE-WG. 17).
Serological and molecular diagnostic methods
Although commercial barley yellow dwarf luteovirus kits were shown to produce positive results with CVEV infected samples, their use is not recommended while the identity of the virus remains in doubt. No molecular methods developed so far.
CONTROL
Use virus-free budwood.
Selected References
Bazan de Segura, C., A. Ferrand B. (1969). Woody gall, its distribution and importance in new and old citrus plantings in Peru. Proc. 1st Intern. Citrus Symp. 3: 1449-1451.
Clark, C. C., J. V.da Graça (2000). Detection of Citrus vein enation virus using cereal yellow dwarf virus ELISA kits. p. 357-359. In J. V. da Graça, R. F. Lee, R. K. Yokomi (eds.) Proc. 14th Conf. Intern. Organization Citrus Virol. IOCV, Riverside, CA.
da Graça, J. V., S. B. Maharaj (1991). Citrus vein enation virus, a probable luteovirus. p. 391-394. In R. H. Brlansky, R. F. Lee, L. W. Timmer (eds.) Proc. 11th Conf. Intern. Organization Citrus Virol.. IOCV, Riverside, CA.
Fraser, L. R. (1958) Virus diseases of citrus in Australia. Proc. Linn. Soc. NSW: 83: 9-19.
Fraser, L. R. (1959) Woody gall, a suspected virus disease of rough lemon and other citrus varieties. Proc. Linn. Soc. NSW 84: 332-336.
Hermoso de Mendoza, A., J. A. Pina, J. F. Ballester-Olmos, L. Navarro. (1993) Persistent transmission of citrus vein enation virus by Aphis gossypii and Myzus persicae. p. 361-362. In P. Moreno, J. V. da Graça, L. W. Timmer (eds.) Proc. 12th Conf. Intern. Organization Citrus Virol. IOCV, Riverside, CA.
Hooper, G. R., H. Schneider (1969) Anatomical investigations of leaf enations on Citrus aurantifolia and C. jambhiri infected with citrus vein enation virus. Phytopathology 57: 1006.
Koizumi, M., A. Sasaki (1980) Protection phenomena against tristeza in trees pre-inoculated with vein enation virus. p. 48-50 In E. C. Calavan, S. M. Garnsey, L. W. Timmer (eds.) Proc. 8th Conf. Intern. Organization Citrus Virol. IOCV, Riverside, CA.
Maharaj, S. B., J. V. da Graça (1988) Observation of isometric virus-like particles associated with citrus vein enation virus-infected citrus and the viruliferous aphid vector Toxoptera citricidus. Phytophylactica 20: 357-360.
Maharaj, S. B., J. V. da Graça (1989) Transmission of citrus vein enation virus by Toxoptera citricidus. Phytophylactica 21: 81-82.
Mali, V. R., K. G. Chaudhuri, S. D. Rane (1975) Vein enation and woody gall virus disease of citrus. FAO Plant Prot. Bull. 23: 190-191.
Vidalakis, G., S. M. Garnsey, J. A. Bash, G. D. Greer, D. J. Gumpf (2004) Efficacy of bioindexing for graft-transmissible citrus pathogens in mixed infections. Plant Dis. 88: 1328-1334.
Wallace, J. M., R. J. Drake (1953) A virus induced vein enation in citrus. Citrus Leaves 33: 22,24.
Wallace, J. M., R. J. Drake (1959) Citrus vein enation. p. 163-165 In J. M. Wallace (ed.) Citrus Virus Diseases, Univ. Calif. Div. Agric. Sci., Berkley, CA.
Wallace, J. M., R. J. Drake (1962) Further studies on the woody gall disease of citrus. Phytopathology 52: 756.
Weathers, L. G. (1960) Yellow-vein disease of citrus and studies of interactions between yellow-vein and other viruses of citrus. Virology 11: 753-764.
Prepared (1975, 1981) by J. M. Wallace
University of California
Riverside CA 92521
(USA)
Revised (2008) by J. V. da Graça
Texas A & M University-Kingsville
Citrus Center
Weslaco TX 78596
(USA)
Click on any image to see larger.
PHOTOS |
LEGENDS AND AUTHORS |
|---|---|
|
Early stage galls on stems of Mexican lime (California, USA) – J. M. Wallace
|
|
|
Rough lemon tree with well developed galls (California, USA) – C. N. Roistacher
|
|
|
Advanced gall growth on rough lemon (right) and a lime (left), the latter showing any sprouts from gall tissue (California, USA) – J. M. Wallace
|
|
|
Woody galls developing at sites of bark punctures on the stem of rough lemon (California, USA) – J. M. Wallace
|
|
|
Bark removed from swellings shown in slide cr 06 showing the undeveloped thorn-like structure (California, USA) – J. M. Wallace
|
|
|
Normal swellings adjacent to thorns on rough lemon (California, USA) – J. M. Wallace
|
|
|
Woody galls on rough lemon rootstock (South Africa) – J. M. Bové
|
|
|
Galls encircling bud-union of tree grown from healthy Valencia bud grafted on to an infected rough lemon rootstock (California, USA) – J. M. Wallace
|
|
|
Bark removed from woody galls to show pitted surface (California, USA) – J. M. Wallace
|
|
|
Vein enations on undersurface of leaves of Mexican lime (California, USA) – J. M. Wallace
|
|
|
Depressions on upper surface of Mexican lime leaves corresponding to enations (California, USA) – J. M. Wallace
|
|
|
Enhancement of yellow vein symptoms in Mexican lime by co-infecting CVEV (California, USA) – L. G. Weathers
|
|
|
Isometric virus-like particles in phloem tissue of CVEV-infected rough lemon (South Africa) – S. B. Maharaj & J. V. da Graça
|
|
|
Virus-like particles purified from rough lemon enations (South Africa) - S. B. Maharaj & J. V. da Graça
|
|
|
Woody galls of unknown etiology on navel orange scion (Turkey) – C. N. Roistacher
|
|
|
Enations on underside of citrus leaf possibly due to insect injury (India) – C. N. Roistacher
|
|
|
Shock symptoms found associated with CVEV in an indexing greenhouse (Spain). – J. A. Pina |